- Mailing Address:
- School of Pharmacy
- University of Wisconsin
- 777 Highland Ave.
- Madison, WI 53705-2222
- Office:
- 5125 Rennebohm Hall
- Phone: 608-265-8491
- Fax: 608-262-5345 Email: lingjun.li@wisc.edu
- Pharmaceutical Sciences Division
- Wisconsin Center for NanoBioSystems
- UW Madison Chemistry Department
- Li Lab Website
- Full List of Publications
- Curriculum Vitae
- Google Scholar
- NCBI

Lingjun Li, PhD
Professor
Vilas Distinguished Achievement Professor of Pharmaceutical Sciences and Chemistry
Charles Melbourne Johnson Distinguished Chair in Pharmaceutical Sciences
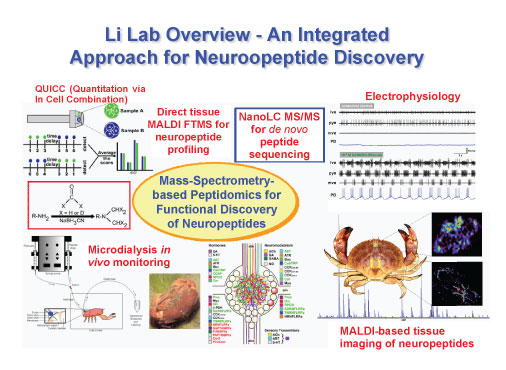
Research in my laboratory focuses on developing and implementing an array of novel mass spectrometry based strategies to answer questions about the most complex and elusive set of signaling molecules, the neuropeptides, and gain new insights into the roles of peptide hormones and neurotransmitters play in the plasticity of neural circuits and behavior. While significant effort has been directed to analytical technique and method development, it is the biomedical importance of understanding the neuropeptidergic system that drives our research to continuously refine and improve the analytical capabilities to address challenging neuroscience problems. A large body of research strongly suggests that an imbalance of chemical messengers is associated with various neurological disorders. However, compared to classical neurotransmitters such as amines, the functional roles of many neuropeptides are still poorly understood. This is, in part, due to the lack of analytical capabilities to measure and identify these low abundance endogenous signaling molecules in a complex microenvironment. Clearly, development of highly sensitive and selective analytical tools for neuropeptide identification and quantitation is in great demand. Specifically, we are interested in understanding the roles that neuropeptides play in food intake, neural network development and response to environmental stresses.
We have chosen to work with a simpler and well-defined crustacean nervous system to both facilitate technology development and address fundamental neuroscience problems related to neuromodulation and network plasticity. We are developing a multi-faceted mass spectrometry-based analytical platform to probe peptidergic signaling with enhanced sensitivity and selectivity. By combining chemical labeling, micro-scale separation (capillary electrophoresis and nanoLC), and tandem mass spectrometry sequencing techniques, we have discovered a large number of novel neuropeptides in crustacean nervous systems. The physiological effects of these new peptides at the cellular and network levels are evaluated in collaboration with neurophysiologists (Eve Marder and Michael Nusbaum) in the field. Furthermore, both mass spectrometric imaging technology and in vivo microdialysis sampling tools have been implemented to follow neuropeptide distribution and secretion with unprecedented details. Finally, a differential display strategy in conjunction with isotopic labeling technique is being developed to allow functional discovery of neuropeptides in response to various physiological changes.
While the technology is developed using crustacean model system as a test-bed, the technology advancement resulting from our research is widely applicable to the large-scale analysis of peptides and proteins in many biological systems, including those of mammalian and humans. Towards this end, we have established several exciting collaborations targeted at neurochemical analysis in more complex systems. These collaborative projects include biomarker discovery in neurodegenerative diseases (with Albee Messing and Judd Aiken), peptide analysis and neuromodulation in mammalian rhythmic neural network (with Jan Marino (Nino) Ramirez), proteomic analysis neuroprotective factors secreted by astrocytes (with Jeffrey Johnson), and proteomic study of dioxin-induced cardiotoxicity in developing zebrafish (with Warren Heideman and Richard Peterson).
These synergistic projects span analytical mass spectrometry, capillary separations, peptide chemistry, bioinformatics, neurochemistry, and neurobiology. The improved analytical method development enables biological discovery, and the emerging biological questions require further advancement of analytical tools.
Background: Lingjun received her BE degree in Environmental Analytical Chemistry from Beijing Polytechnic University and a PhD degree in Analytical Chemistry/Biomolecular Chemistry from University of Illinois at Urbana-Champaign. She did three-way postdoctoral research at the Pacific Northwest National Laboratory, Brandeis University, and University of Illinois before joining the School of Pharmacy faculty in 2002. She currently holds joint appointments in School of Pharmacy and Department of Chemistry at UW-Madison. Her research interests are in analytical neurochemistry, neuroproteomics and biological mass spectrometry.
Professional Interests: Drug Action and Drug Discovery
Education:
- BE 1992 Env. Analyt. Chem. - Beijing Polytechnic University
- PhD 2000 Analyt & Biomol Chem - University of Illinois at Urbana-Champaign (UIUC)
- Postdoc 2002 Analyt Chem/NeuroSci - Pacific Northwest National Laboratory, Brandeis University, UIUC
- G. Li, K. DeLaney, K., and L. Li* (2019). Molecular basis for chirality-regulated Abeta self-assembly and receptor recognition revealed by ion mobility-mass spectrometry. Nature Communications. Nov 6;10(1):5038. doi: 10.1038/s41467-019-12346-8.
- G. Li, F. Ma, Q. Cao, Z. Zheng, K. DeLaney, R. Liu, and L. Li* (2019). Nanosecond photochemically promoted click chemistry for enhanced neuropeptide visualization and rapid protein labeling. Nature Communications. Oct 16;10(1):4697. doi: 10.1038/s41467-019-12548-0. Recommended by Faculty 1000, and featured as Editor’s Highlights
- G. Li, A. Phetsanthad, M. Ma, Q. Yu, A. Nair, Z. Zheng, K. DeLaney, F. Ma, S. Hong, and L. Li* (2020). Expedited structural mapping of glycoprotein transferrin sialylation associated with Alzheimer’s disease. Chemical Science. Under revision.
- Frost, Y. Feng, and L. Li* (2020). 21-plex DiLeu isobaric tags for high-throughput quantitative proteomics. Analytical Chemistry. Under revision.
- K. DeLaney, A. Phetsanthad, and L. Li* (2020). Advances in high-resolution MALDI mass spectrometry for neurobiology. Mass Spectrometry Reviews. Invited contribution for a Special Issue honoring Alan Marshall. Acceptance pending minor revision.
- L.E. Atkinson, Y. Liu, F. McKay, E. Vandewyer, C. Viau, A. Irvine, B.A. Rosa, Z. Li, N.J. Marks, A.G. Maule, M. Mitreva, I. Beets, L. Li, and A. Mousley* (2020). Extrasynaptic volume transmission: a novel route for neuropeptide signaling in nematodes. Submitted.
- D.G. Delafield and L. Li* (2020). Recent advances in analytical approaches for glycan and glycopeptide quantitation. Molecular and Cellular Proteomics. Invited review for the Special Issue on Recent Advances in Glycoproteomics and Glycomics.
- X. Zhong, D. Frost, Q. Yu, M. Li, T.-J. Gu, and L. Li* (2020). Mass defect-based DiLeu tagging for multiplexed data-independent acquisition. Analytical Chemistry. Under review.
- X. Zhong, C.B. Lietz, X. Shi, A.R. Buchberger, D.C. Frost, and L. Li* (2020). Highly multiplexed quantitative proteome and phosphoproteomic analyses in vascular smooth muscle cell dedifferentiation. Analytica Chimica Acta. Under review.
- J. Zhu, J. Huang, J. Zhang, Z. Chen, Y. Lin, G. Grigorean, L. Li, S. Liu, A. Singal, N. Parikh, and D. Lubman* (2020). Glycopeptide biomarkers in serum haptoglobin for hepatocellular carcinoma detection in patients with non-alcoholic steatohepatitis. Journal of Proteome Research. Under revision
- Q. Yu, X. Zhong, B. Chen, Y. Feng, M. Ma, C.A. Diamond, J.S. Voeller, M. Kim, K.B. DeSante, C.M. Capitini, N.J. Patel, M.L. Hoover-Regan, M.J. Burke, K. Janko, D.M. Puccetti, C. Ikonomidou, and L. Li* (2020). Isobaric labeling strategy utilizing 4-plex N,N-dimethyl leucine (DiLeu) tags reveals proteomic changes induced by chemotherapy in cerebrospinal fluid of children with B-cell acute lymphoblastic leukemia. Journal of Proteome Research. Under revision.
- P. Wei, L. Hao, S. Thomas, A.R. Buchberger, L. Steineke, P.C. Marker, W.A. Ricke, and L. Li* (2020). Urinary amine metabolomics characterization of a hormone-induced urinary obstruction mouse model with custom 12-plex isobaric DiLeu labeling. Journal of the American Society for Mass Spectrometry. Under review.
- G. Lu, X. Xu, G. Li, H. Sun, N. Wang, Y. Zhu, N. Wan, Y. Shi, G. Wang, L. Li, H. Hao, and H. Ye* (2020). Subresidue-resolution footprinting of ligand-protein interactions by carbene chemistry and ion mobility-mass spectrometry. Analytical Chemistry. 92, 947-956.
- K. DeLaney and L. Li* (2020). Capillary electrophoresis coupled to MALDI mass spectrometry imaging with large volume sample stacking injection for improved coverage of C. borealis neuropeptidome. Analyst, 145, 61-69. DOI: 10.1039/C9AN01883B. Featured on the back cover of the journal
- N.T. Chan, P. Liu, J. Huang, Y. Wang, I. Ong, L. Li, and W. Xu* (2020). Transcriptional elongation machinery controls vulnerability of breast cancer cells to PRC2 inhibitors. bioRxiv
- R. Liu, Y. Huang, H. Xu, Y. Zheng, Y. Liu, S. Han, M. Yang, Y. Xie, K. Wang, J.A. Duan, and L. Li* (2019). A strategy for identifying species-specific peptide biomarkers in deer-hide gelatin using untargeted and targeted mass spectrometry approaches. Analytica Chimica Acta, 1092, 32-41.
- Cao, Q. Yu, Y. Liu, Z. Chen, and L. Li* (2020) Signature-ion triggered mass spectrometry approach enabled discovery of N- and O-linked glycosylated neuropeptides in the crustacean nervous system. Journal of Proteome Research, DOI: 10.1021/acs.jproteome.9b00525
- L. Hao, Y. Zhu, P. Wei, J. Johnson, A. Buchberger, D. Frost, W.J. Kao, and L. Li* (2019). Metandem: An online software tool for mass spectrometry-based isobaric labeling metabolomics. Analytica Chimica Acta, 1088, 99-106.
- K. DeLaney, W. Cao, Y. Ma, M. Ma, Y. Zhang, and L. Li* (2020). PRESnovo: Prescreening prior to de novo sequencing to improve accuracy and sensitivity of neuropeptide identification. Journal of the American Society for Mass Spectrometry. In press. https://doi.org/10.1021/jasms.0c00013 Invited contribution to the Special Focus Issue on Bioinformatics in Omics Research, honoring John Yates on his winning the 2019 John B. Fenn Award for a Distinguished Contribution in Mass Spectrometry.
- G. Li, D.G. Delafield, and L. Li* (2020). Improved structural elucidation of peptide isomers and their receptors using advanced ion mobility-mass spectrometry. Trends in Analytical Chemistry (TrAC). 124, 115546. https://doi.org/10.1016/j.trac.2019.05.048. Invited contribution for special issue on Ion Mobility Spectrometry: From Fundamentals to Applications.
- Y. Lv, J. Fu, Q. Jia, H. Dong, S. Han, L. Li*, L. He (2020). A liquid chromatography tandem mass spectrometry based label-free quantification method for assessment of allergen-induced anaphylactoid reactions. Journal of the American Society for Mass Spectrometry. 31, 856-863.
- D.M. Tilley, C.B. Lietz, D.L. Cedeno, C.A. Kelley, L. Li, R. Vallejo (2020). Proteomic modulation in the dorsal spinal cord following spinal cord stimulation therapy in an in vivo neuropathic pain model. Neuromodulation: Technology at the Neural Interface. In press, https://doi.org/10.1111/ner.13103
- A.R. Buchberger, N.Q. Vu, J. Johnson, K. DeLaney, and L. Li* (2020). A simple and effective sample preparation strategy for MALDI-MS imaging of neuropeptide changes in the crustacean brain due to hypoxia and hypercapnia stress. Journal of the American Society for Mass Spectrometry. In press. https://doi.org/10.1021/jasms.9b00107. Featured as a supplemental cover.
- P. Wei, C. Keller, and L. Li* (2020). Neuropeptides in gut-brain axis and their influence on host immunity and stress. Computational and Structural Biotechnology Journal. 18, 843-851. Invited contribution.
- S. Thomas, L. Hao, K. DeLaney, D. McLean, L. Steinke, P.C. Marker, C.M. Vezina, L. Li*, and W.A. Ricke (2020). Spatiotemporal proteomics reveals the molecular consequences of hormone treatment in a mouse model of lower urinary tract dysfunction. Journal of Proteome Research. 19, 1375-1382.
- A.R. Buchberger, C.S. Sauer, N.Q. Vu, K. DeLaney, and L. Li* (2020). Temporal study of the perturbation of crustacean neuropeptides due to severe hypoxia using 4-plex reductive demethylation. Journal of Proteome Research. 19, 1548-1555. Featured as a supplemental cover.
- M. Vestal, L. Li, E. Dobrinskikh, Y. Shi, B. Wang, X. Shi, S. Li, C. Vestal, and K. Parker. (2019). Rapid MALDI-TOF molecular imaging: instrument enhancements and their practical consequences. Journal of Mass Spectrometry. 2019 Jul 17. doi: 10.1002/jms.4423.
- Feng, B. Chen, Q. Yu, and L. Li* (2019). Identification of double bond position isomers in unsaturated lipids by m-CPBA epoxidation and mass spectrometry fragmentation. Analytical Chemistry. 91, 1791-1795.
- M. Ruiz, Y. Yang, C.A. Lochbaum, D.G. Delafield, J.J. Pignatello, L. Li, J.A. Pedersen* (2019). Peroxymonosulfate oxidizes amino acids in water without activation. Environmental Science and Technology. 53, 10845-10854.
- F. Ma, D.M. Tremmel, Z. Li, C.B. Lietz, S.D. Sackett, J.S. Odorico, L. Li* (2019). In-depth quantification of extracellular matrix proteins from human pancreas. Journal of Proteome Research. 18, 3156-3165.
- X. Zhong, D.C. Frost, and L. Li* (2019). High-resolution enabled 5-plex mass defect-based N,N-dimethyl leucine tags for quantitative proteomics. Analytical Chemistry. 91, 7991-7995.
- L. Hao, S. Thomas, T. Greer, C.M. Vezina, S. Bajpai, A. Ashok, A.M. De Marzo, C.J. Bieberich, L. Li*, and W.A. Ricke (2019). Quantitative proteomic analysis of a genetically induced prostate inflammation mouse model via custom 4-plex DiLeu isobaric labeling. American Journal of Physiology-Renal Physiology. 316, F1236-F1243.
- DeLaney and L. Li* (2019). Data independent acquisition mass spectrometry method for improved neuropeptidomic coverage in crustacean neural tissue extracts. Analytical Chemistry. 91, 5150-5158.
- Q. Cao, Y. Wang, B. Chen, F. Ma, L. Hao, G. Li, C. Ouyang, and L. Li* (2019). Visualization and identification of neurotransmitters in crustacean brain via multifaceted mass spectrometric approaches. ACS Chemical Neuroscience. 10, 1222-1229. Featured on the journal cover.
- K. DeLaney, C.S Sauer, N.Q. Vu, and L. Li* (2019). Recent advances and new perspectives in capillary electrophoresis-mass spectrometry for single cell “omics”. Molecules 24, 42. https://doi.org/10.3390/molecules24010042
- S.D. Sackett, D.M. Tremmel, F. Ma, A.K. Feeney, R.M. Maguire, M.E. Brown, Y. Zhou, X. Li, C. O’Brien, L. Li, W.J. Burlingham, J.S. Odorico* (2018). Extracellular matrix scaffold and hydrogel derived from decellularized and delipidized human pancreas. Scientific Reports. 8, 10452. https://doi.org/10.1038/s41598-018-28857-1.
- F. Liu, F. Ma, Y. Wang, L. Hao, H. Zeng, C. Jia, Y. Wang, P. Liu, I.M. Ong, B. Li, G. Chen, J. Jiang, S. Gong, L. Li, and W. Xu* (2018). PKM2 methylation by CARM1 activates aerobic glycolysis to promote tumorigenesis. Cancer Research. 78, 13 supplement, 2418.
- A.E. Turco, S. Thomas, L.K. Crawford, W. Tang, R.E. Peterson, L. Li, W.A. Ricke, and C.M. Vezina* (2020). In utero and lactational 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) exposure exacerbates urinary dysfunction in hormone-treated C57BL/6J mice through a non-malignant mechanism involving proteomic changes in the prostate that differ from those elicited by testosterone and estradiol. American Journal of Clinical Experimental Urology. 8, 59-72.
- X. Yan, L. Li, and C. Jia (2019). Discovery of missing methylation sites on endogenous peptides of human cell lines. Journal of The American Society for Mass Spectrometry, 30, 2537-2547.
- Y. Cui, K. Yang, D.N. Tabang, J. Huang, W. Tang, and L. Li* (2019). Finding the sweet spot in ERLIC mobile phase for simultaneous enrichment of N-glyco and phosphopeptides. Journal of The American Society for Mass Spectrometry, 30, 2491-2501. Invited contribution to Focus Issue in Honor of Benjamin A. Garcia, Recipient of the 2018 ASMS Biemann Medal